278 patents from 49 companies…an increase of 123 cases compared to 2021
[Newsmp] Last year, the number of patents registered in the Ministry of Food and Drug Safety (MFDS) reached its highest level since 2014.
According to the MFDS information platform (https://nedrug.mfds.go.kr/), a total of 278 patents were registered in 2022, up 123 cases from the previous year.
It is the first time since 2014 that the number of patents registered has exceeded 250 per year.
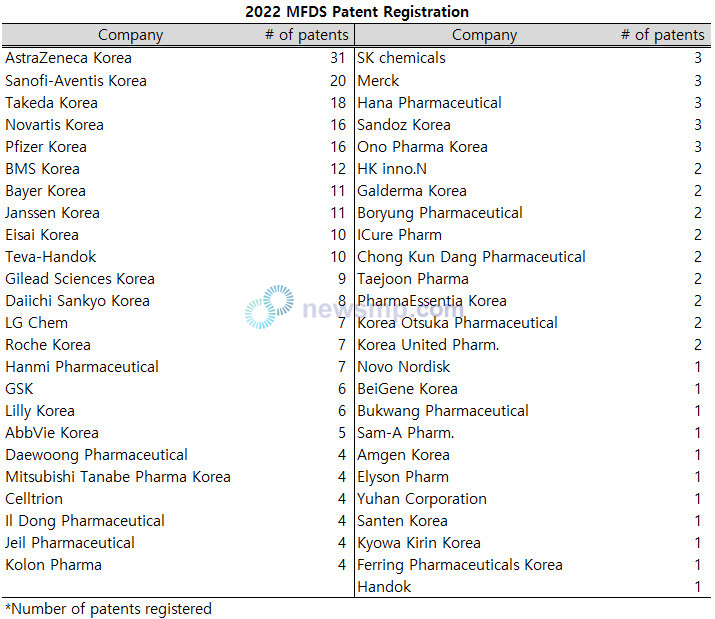
A total of 49 companies registered patents, and 10 of them registered more than 10 patents.
AstraZeneca Korea took the lead with 31 cases, the only one to exceed 30 cases, followed by Sanofi-Aventis Korea with 20 cases.
Takeda Korea had 18 cases, Novartis Korea 16 cases and Pfizer Korea 15 cases.
BMS Korea recorded 12 cases, Bayer Korea and Janssen Korea 11 cases, and Eisai Korea and Teva-Handok 10 cases, exceeding double digits.
Moreover, Gilead Sciences Korea had nine cases, Daiichi Sankyo Korea eight cases, LG Chem, Roche Korea and Hanmi Pharmaceutical seven cases, GSK and Lilly Korea six cases, and AbbVie Korea five cases.
Daewoong Pharmaceutical, Mitsubishi Tanabe Pharma Korea, Celltrion, Il Dong Pharmaceutical, Jeil Pharmaceutical and Kolon Pharma registered four cases, and SK chemicals, Merck, Hana Pharmaceutical, Sandoz Korea and Ono Pharma Korea three cases.
HK inno.N, Galderma Korea, Boryung Pharmaceutical, ICure Pharm, Chong Kun Dang Pharmaceutical, Taejoon Pharma, PharmaEssentia Korea, Korea Otsuka Pharmaceutical and Korea United Pharm. recorded two cases each.
Novo Nordisk, BeiGene Korea, Bukwang Pharmaceutical, Sam-A Pharm., Amgen Korea, Elyson Pharm, Yuhan Corporation, Santen Korea, Kyowa Kirin Korea, Ferring Pharmaceuticals Korea and Handok had one case.


