Among the 1151 drugs listed in the Korean version of Orange Book last year, Statin plus Ezetimibe combination drugs accounted for more than 300 cases.
According to the Ministry of Food and Drug Safety’s information platform (https://nedrug.mfds.go.kr/), the most registered in the K-Orange Book based on its main ingredients was Ezetimibe drugs, 189 cases out of 1151 cases.
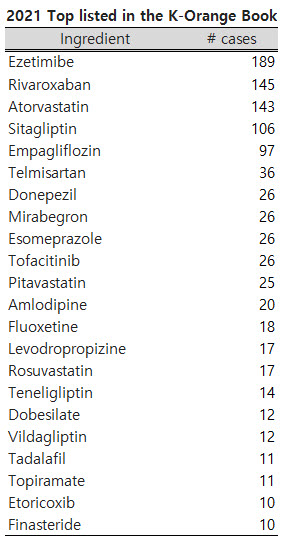
Following Ezetimibe, Rivaroxaban had 145 cases, Atorvastatin 143 cases, and Sitagliptin 106 cases, exceeding 100 cases.
Among them, most of the Ezetimibe and Atorvastatin drugs were listed as combinations, but either Ezetimibe or Atorvastatin was listed as the main ingredient, and in fact, there were more than 300 cases of Atorvastatin plus Ezetimibe combinations.
Twenty-five cases of Pitavastatin and 17 cases of Rosuvastatin were listed, and many of them were also Ezetimibe combinations.
Following the four ingredients that exceeded 100, the Empagliflozin was close to 100 by 97 cases.
Telmisartan had 36 cases, and Donepezil, Mirabegron, Esomeprazole, and Tofacitinib were 26 cases.
There were 25 cases of Pitavastatin, 20 cases of Amlodipine, 18 cases of Fluoxetine, and 17 cases of Levodropropizine and Rosuvastatin.
Moreover, Teneligliptin recorded 14 cases, Dobesilate and Vildagliptin 12 cases, Tadalafil and Topiramate 11 cases, and Etoricoxib and Finasteride 10 cases.


