[Newsmp] The number of new entries in the patent list of the Ministry of Food and Drug Safety significantly decreased, not exceeding 100 cases during the last six months.
According to the MFDS information platform (https://nedrug.mfds.go.kr/), the Ministry of Food and Drug Safety's patent list for the previous first half of the year saw a total of 29 companies registering 95 patents. This number reflects a considerable decline of 37 cases or a 28% decrease compared to the same period in the previous year, which recorded 132 cases.
The number of newly registered patents increased from 90 cases in 2018 to 118 cases in 2019, marking a recovery to a three-digit figure in four years since 2015.
Despite the COVID-19 pandemic, the number of patent registrations expanded to 134 cases in 2020 but experienced a sharp drop to 58 cases in 2021.
In 2022, there was another surge to 132 cases, reaching a level similar to 2020, but in the first half of the year, it decreased by 37 cases, dropping to double digits.
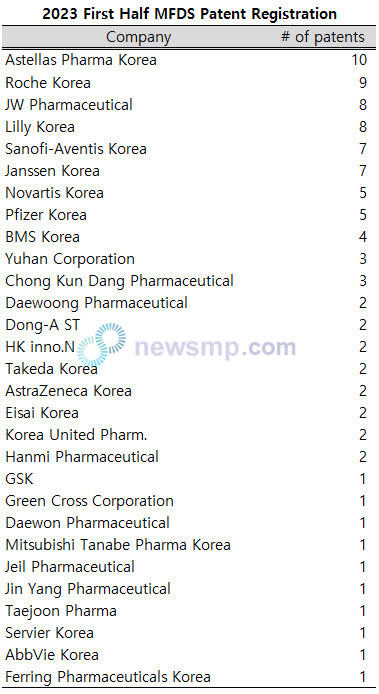
Among the 29 companies, Astellas Pharma Korea recorded a total of 10 patents, achieving a double-digit number.
Roche Korea registered 9 patents, while JW Pharmaceutical and Lilly Korea secured 8 patents each, followed by Sanofi-Aventis Korea and Janssen Korea with 7 patents, and Novartis Korea and Pfizer Korea with 5 patents.
Moreover, BMS Korea recorded 4 patents, Yuhan Corporation and Chong Kun Dang Pharmaceutical obtained 3 patents each, and Daewoong Pharmaceutical, Dong-A ST, HK inno.N, Takeda Korea, AstraZeneca Korea, Eisai Korea, Korea United Pharm., and Hanmi Pharmaceutical were listed with 2 patents each.
GSK, Green Cross Corporation, Daewon Pharmaceutical, Mitsubishi Tanabe Pharma Korea, Jeil Pharmaceutical, Jin Yang Pharmaceutical, Taejoon Pharma, Servier Korea, AbbVie Korea, and Ferring Pharmaceuticals Korea also made the list by each contributing one patent.


