Imfinzi (Durvalumab, AstraZeneca) demonstrated a 5-year overall survival rate of 42.9% for patients with unresectable stage 3 non-small cell lung cancer (NSCLC).
Imfinzi has become a new standard therapy in this field by lowering the risk of death through consolidation therapy for one year after concurrent chemoradiation therapy (CRT) in the phase 3 PACIFIC study conducted on patients with unresectable stage 3 NSCLC.
Earlier in 2018, the International Association for the Study of Lung Cancer announced that Imfinzi reduced the risk of death by 32% (Overall Survival, OS HR=0.68, P=0.00251) and the risk of disease progression or death by 52% (Progression-Free Survival, PFS HR=0.52, P<0.0001) compared to placebo regardless of PD-L1 expression.
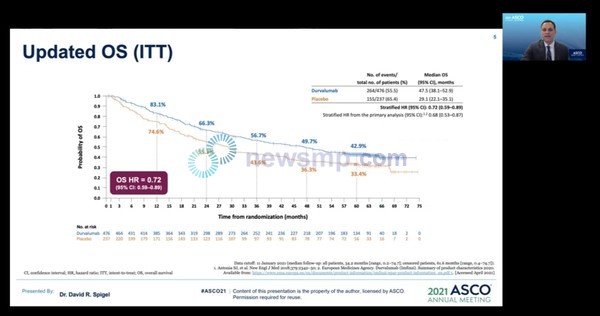
Meanwhile, five-year follow-up data of phase 3 PACIFIC trials was released at ASCO Annual Meeting, which opened on the 4th.
According to David Spigel, MD, chief scientific officer at the Sarah Cannon Research Institute, the five-year overall survival rate of 42.9% for Imfinzi-treated and 33.4% for placebo.
The median OS was 47.5 months for Imfinzi and 29.1 months for placebo groups, and the risk of death in the Imfinzi group was lower by 28% (HR=0.72).
Following a maximum treatment course of a year, 33.1% of patients treated with Imfinzi had not progressed 5 years compared to 19% for placebo.
Median 5-year PFS in the Imfinzi was 16.9 months and 5.6 months in the placebo (HR=0.55), and the risk of disease progression or death was lower by 45%.
Spigel said, “This trial has once again set a new precedent in the treatment of patients with unresectable Stage III non-small cell lung cancer.”


